
Key Points
MaavRx is developing a one-time treatment to restore lifelong constant production of L-DOPA in patients with advanced PD.
We believe this will be more effective, safer, more convenient and cheaper than current treatment.
Strong preclinical proof of efficacy, safety in the classic primate model of PD.
FDA agreed preclinical proof of concept data and outline clinical development.
Seeking Series A funding.
Predicted sales $2 billion/ annum.
Investment already significantly derisked.

The Parkinson's Disease Crisis

12 Million Patients, 25 Million by 2025
Prevelence is growing faster than the ageing population.
Affects 2% of population over 60 year old
The second most common neurodegenerative condition after Alzheimer’s
Motor Symptoms
Difficulty moving (slowness, rigidity, “freezing”, tremor)
Leading to progress loss of independence
$50B annual cost in the USA alone
$25B in direct medical costs
$25B in lost earnings of patients and caregivers
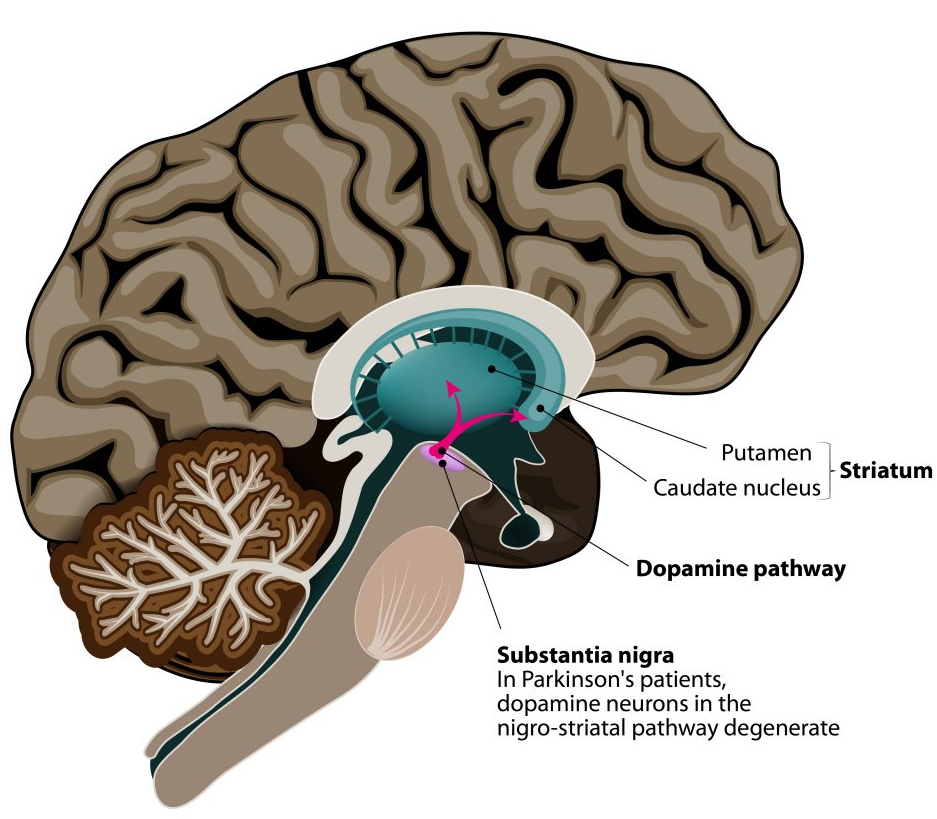
The central role of L-DOPA
Parkinson’s disease causes increasing difficulty with movement and loss of independence.
Due to loss of cells delivering L-DOPA and dopamine to the brain putamen.
Symptoms develop when level of L-DOPA and dopamine fall below 20% of normal.
L-DOPA is the most effective treatment. It is converted to dopamine in the brain.
Almost every patient will be treated with L-DOPA as their disease progresses.
Oral L-DOPA
Oral L-DOPA taken several times a day is very effective for the first years of treatment.
But variable absorption and a short half-life results in fluctuating erratic levels of L-DOPA in the brain.
Over time this causes problems.
By 5 years 50-80% of patient treated with oral L-DOPA develop erratic efficacy (on/off variation) and new abnormal movements (dyskinesia)

Dyskinesia following several years of oral L-DOPA
Continuous subcutaneous infusion reduces peak/trough fluctuation
Improved control
- Increased “on-time” by 1.95 hours/day versus oral L-DOPA
- 49% Reduction in “off-time” vs oral L-DOPA (p<0.001)
Reduced dyskinesia
- 48% reduction in dyskinesia disability vs oral (p=0.002)
- 78% reduction in dyskinesia pain (p=0.02)
Approved by FDA, EMA, MHRA etc.
- Marketed by AbbVie as Vyalev in USA, Produodopa in Europe
- Analysts predict sales for continuous infusion of L-DOPA based products will reach $2 billion by 2030
Before continuous infusion
During continuous infusion
Continuous infusion - better control - but far from perfect

- Still variable absorption depending on site, skin reactions and competitive transport across blood brain barrier.
Need to wear pump 24/7/365
Change infusion site every 1-2 days
High cost ($50K/year in USA)
Difficult for patients lacking dexterity
52% of patients give up within a year


MaavRx and better life for patients with advanced Parkinson's disease
How MaavRx works
The motor symptoms of PD are due to the progressive loss of a cluster of cells supplying L-DOPA and dopamine to the brain putamen.
The neurons in the putamen remain healthy but do not function normally without L-DOPA
MaavRx uses proven AAV gene therapy technology to enable neurons in the putamen to make their own L-DOPA
We use an AAV5 vector to deliver the two genes (TH and GCH1) into the neurons.
Because these neurons survive for the lifetime of the patient, the benefit is expected to be lifelong

AAV5 vector particles deliver the genes to neurons in the putamen to enable them to produce constant non-fluctuation levels of L-DOPA

What are the expected advantages
One-time treatment for lifelong effect.
Constant levels of L-DOPA production for improved movement with no or reduced dyskinesia
No pump. No skin reactions.
Lower cost than continuous infusion.

What is the evidence MaavRx will work
Efficacy of prototype confirmed in classic (6OHDA) rat model of Parkinson’s disease.
Efficacy of prototype confirmed in classic (MPTP) primate model of disease.
Evidence that effect persist for 15 years in primate brain.
MaavRx AAV uses same vector, serotype, promoter and transgenes as prototype and with increase potency in vitro and strong expression throughout the putamen of large animals.
Preclinical proof of concept agreed by FDA – No need for further large animal efficacy studies

Single injection of 9E10 and 9E11 gene copies/animal cause significant continued improvement versus vehicle and low dose in primate model of PD. (Readministration of 9E10 also results in efficacy in animals previously treated with subtherapeutic dose)

What is the evidence MaavRx will be safe
Safety of prototype confirmed in GLP acute and chronic toxicology studies in rats and primates. No red flags.
No evidence of increased dyskinesia at highest doses in primate studies.
Similar technology using AAV vector to deliver a different gene into the same area of brain in children already approved by FDA and EMA (Upstaza for AADC deficiency)
Same technology using the same AAV serotype to deliver a different gene into the same area of brain confirmed to be well tolerated and effective in Huntington’s disease.
How easy is MaavRx to administer?
An outpatient MRI of the head is conducted to plan the infusion of MaavRx
MaavRx if infused locally via cannula into the right and left putamen.
Once the patient is prepared and positioned it takes about 10 minutes to insert the cannulas under local or general anaesthetic.
The patient is then moved to a recovery room and the infusion occurs while the patient is awake.
The cannulas are removed under local anaesthetic.
The patient goes home after overnight observation.
MaavRx Ltd plans to use Neurochase’s elegant cannula system to reduce cost and time for delivery

A greatly de-risked opportunity
Preclinical Safety and Efficacy
- Dose related efficacy in multiple published studies in rat and primate models of PD
- Expression persists for 15 years in primates
- Well tolerated. No dyskinesia. No red flags on acute and chronic tox. studies in rats and primates
- Low dose, local administration – avoids AAV high dose systemic toxicity
Relevant Human Data
- Ample clinical data demonstrating the benefit of non-fluctuating L-DOPA
- Same AAV serotype, also administered to the putamen in the same way: confirmed safe in patients (Huntington’s disease)
Regulatory and IP
- Regulatory pathway and clinical study design defined by EMA/FDA approval of continuous subcutaneous L-DOPA prodrug.
- Same AAV technology, same target (putamen), same delivery already approved by EMA/FDA (“Upstaza”)
- Positive “Interact Meeting” with FDA
- Approved patents USA and Europe
Commercial and Health Economics
- Continuous subcutaneous infusion of L-DOPA at $50,000 / patient / year is reimbursed by US Insurers and UK NICE
- Factory gate CoG <$500/ patient (Allows attractive commercial margin)
- Administration employs on techniques already used for DBS in >7000 centres and >100,000 patients